

|
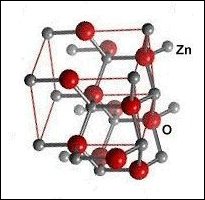
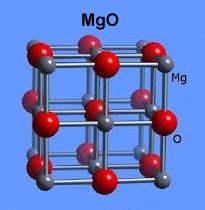
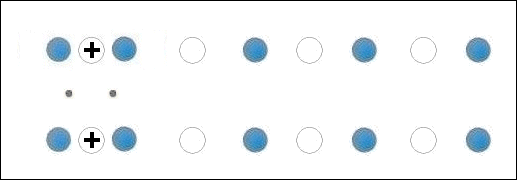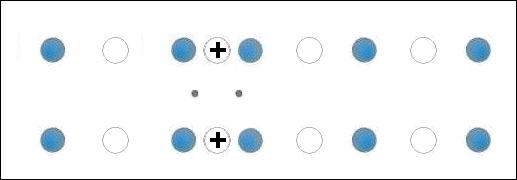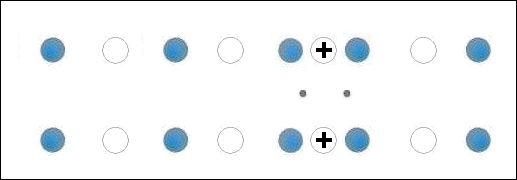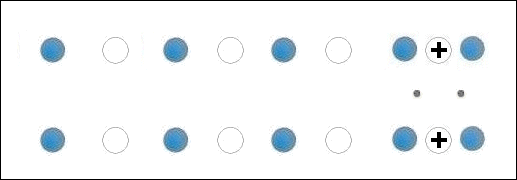
Though the atomic structures of ZnO and MgO (shown at left) are completely different, when they are blended together oxygen gets sandwiched between disparate-weight metals, both of which have +2,+1 oxidation states. This is the critical arrangement that appears to facilitate high-temperature superconductivity. 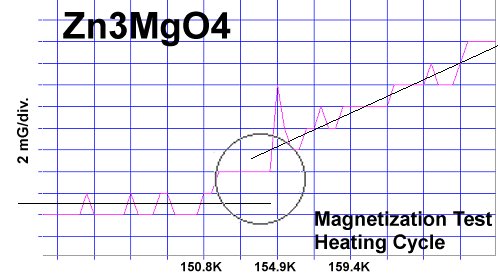
|
A number of +2,+1 metals have already been found to be facilitators of HTSC. These include Cu, Mg, Sr, Ca and Cd. As such, the discovery of Zn superconductivity buttresses the working theory that states: When +2,+1 metals of different atomic weights are positioned on opposite sides of an oxygen atom, periodic compression from lattice vibrations forces a momentary valency shift in the metal from +2 to +1, causing holes to appear at the oxygen site. That positive oxygen ion then acts as a mediator, encouraging electrons to pair up, producing a superconductive state. See the below animation.
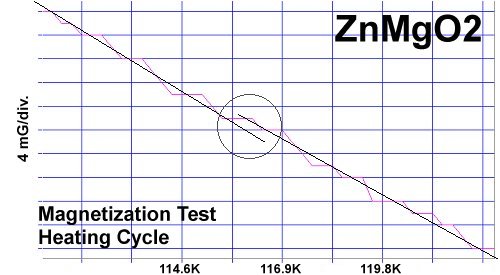
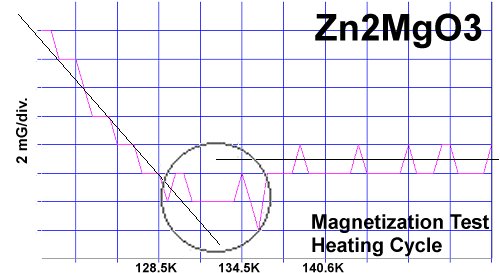
The chemical precursors were pelletized at 60,000 PSI and sintered for 10 hours at 880C. They were then annealed for 10+ hours at 500C in flowing O2. Temperature was determined using an Omega type "T" thermocouple and precision OP77 DC amplifier. The magnetometer employed twin Honeywell SS94A1F Hall-effect sensors with a tandem sensitivity of 50 mv/Gauss.
RESEARCH NOTES: These oxides can be strongly hygroscopic. All tests should be performed immediately after annealing.
RE-PUBLICATION NOTICE: Elsevier Publishing, dba Elsevier Science, as well as Morris Communications, both print and broadcast divisions, are specifically prohibited from re-publishing any part of this news story.
E. Joe Eck
© 2016 Superconductors.ORG
All rights reserved.
 BACK to "News" page at Superconductors.ORG
BACK to "News" page at Superconductors.ORG