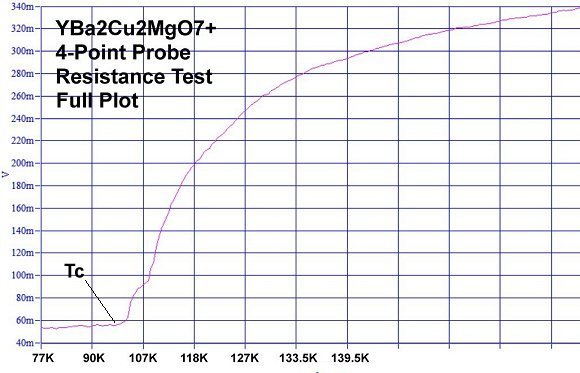
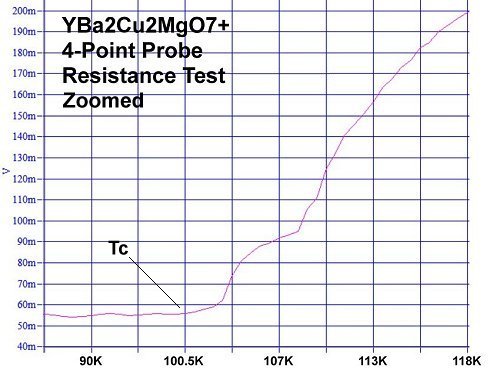


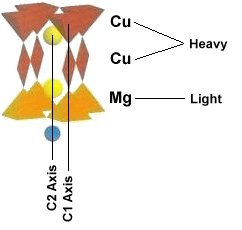
Superconductors.ORG herein reports, through the application of planar weight disparity (PWD), substituting magnesium into every 3rd copper atomic site in YBCO will increase its Tc from 91 Kelvin to 100 Kelvin. The resulting YBa2Cu2MgO7+ compound now has planar weight disparity in both the C1 and C2 axes (see graphic below left). In conventional YBCO (Y123) there exists PWD in the C2 axis only - between the heavy barium atoms on top (yellow) and the lighter yttrium atom below (blue). With this improved formulation PWD now exists in the C1 axis as well, between the heavier copper atoms on top (center red) and the lighter magnesium atom below (center orange). The new YBCO-Mg hybrid, like its progenitor, remains non-toxic and inexpensive to manufacture.
 |
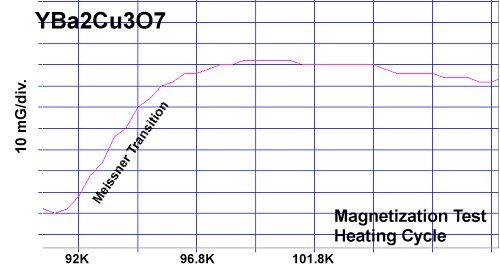
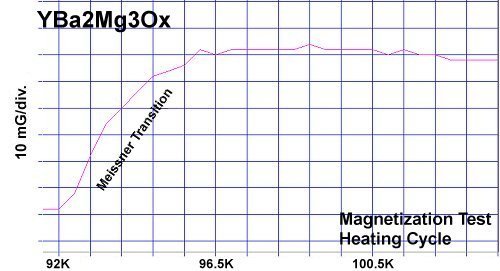
In May of 2016 it was found that magnesium (Mg) was a viable substitute for copper in many of the known superconducting copper-oxides. Below, for example, YBMO produced almost exactly the same response curve as YBCO. Both materials displayed diamagnetic Tc's around 91 - 92K.
|
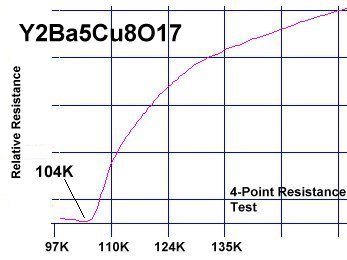
Magnesium, like copper, has +1, +2 oxidation states with an effective ionic radius that's within 2% of copper's EIR. So it was felt YBa2Cu2MgO7+ would likely retain the 1212C structure. The resulting 100K Tc confirmed this. The most unusual aspect of the YBCO-Mg resistance plot is its gradual decline with temperature. It is similar to the plot of Y258, as seen below. YBCO-Mg also has a transition "bumper" just above Tc. (See plots at page top.)
The below list of chemical precursors were stoichiometrically mixed and pelletized. The pellet was then sintered for 11 hours at 900C and annealed for 10+ hours at 500C in flowing O2. Testing temperatures were determined using a type "T" thermocouple (Omega) and precision OP77 DC amplifier (Analog Devices). The material was sufficiently hard for leads to be bonded to the pellet with 8331 silver epoxy (MG Chemicals). Multiple data plots suggest the volume fraction is high.
RESEARCH NOTES: The copper-oxides can be strongly hygroscopic. All tests should be performed immediately after annealing.
E. Joe EckRE-PUBLICATION NOTICE: Elsevier Publishing, dba Elsevier Science, as well as Morris Communications, are specifically prohibited from re-publishing any part of this news story.
 BACK to "News" page at Superconductors.ORG
BACK to "News" page at Superconductors.ORG