HTS Theory Gets A Boost from Rare Earth Metals


|
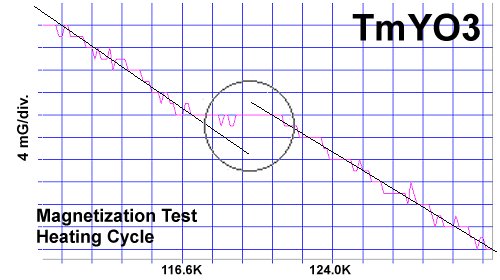
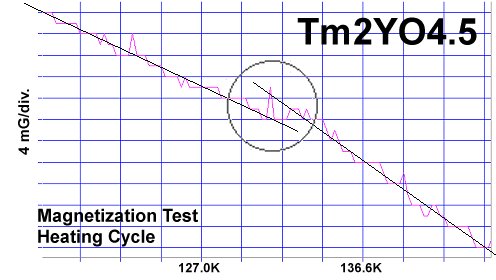
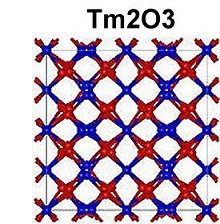
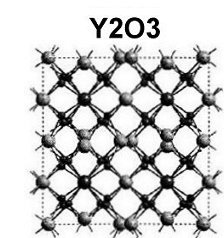
The magnetization plots at page top show Meissner transitions of about 8 milligauss for TmYO3 and 4-5 milligauss for Tm2YO4.5. The below plot of Tm3YO6, which appeared as a minority phase in the other two formulations, shifts about 3-4 milligauss. The transition temperatures (Tc) were 117K, 130K and 140K respectively. Though the volume fraction is low, the evidence is unambiguous, appearing in over a half-dozen tests of each material.[2] Lines have been drawn through the noise to approximate the average of the data points.[3] 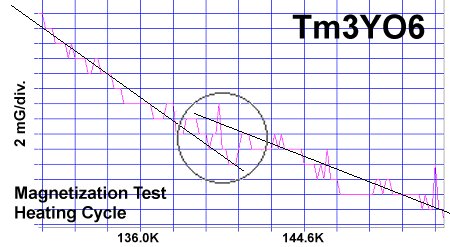
|
The chemical precursors were pelletized at 60,000 PSI and sintered for 10 hours at 880C. They were then annealed for 10+ hours at 500C in flowing O2. Temperature was determined using an Omega type "T" thermocouple and precision OP77 DC amplifier. The magnetometer employed twin Honeywell SS94A1F Hall-effect sensors with a tandem sensitivity of 50 mv/Gauss.
[1] Prior research by ORNL and the University of Rome has determined that high-temperature superconductivity "arises from the oxygen ions". See: "High-Tc Superconductivity at the Interface between the CaCuO2 and SrTiO3 Insulating Oxides" Phys. Rev. Lett. 115, 147001 – Published 28 September 2015. dx.doi.org/10.1103/PhysRevLett.115.147001RESEARCH NOTES: These oxides can be strongly hygroscopic. All tests should be performed immediately after annealing.
RE-PUBLICATION NOTICE: Elsevier Publishing, dba Elsevier Science, as well as Morris Communications, both print and broadcast divisions, are specifically prohibited from re-publishing any part of this news story.
E. Joe Eck
© 2016 Superconductors.ORG
All rights reserved.
 BACK to "News" page at Superconductors.ORG
BACK to "News" page at Superconductors.ORG