

Superconductors.ORG herein reports synthesis of the first room-temperature superconductor that does not contain any barium. Exhibiting a critical transition temperature (Tc) near 33 Celsius, the chemical formula of this new material is Sn4Sb4MnCu8Ox. The importance of this discovery lies in its simple quasi-infinite-layer structure. Such an atomic structure would lend itself to mass-production via "molecular engineering."
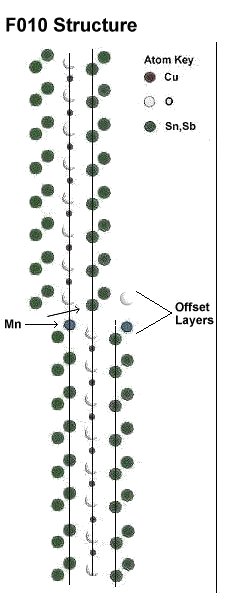
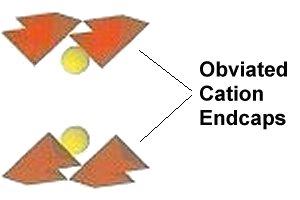
In order to attain room temperature superconductivity in a copper-oxide, a high planar weight ratio (PWR) must be established across the oxygen layers that separate the anion from the cation. In the first 33 RT superconductors that were discovered, this was accomplished using barium and tetragonal copper-oxide "endcaps" (shown below right). To get a high PWR without BaCuO endcaps, a theoretical F010 structure was conceived that has an offset between unit cells (shown below left). The high dielectric constant alloy SnSb (K=147) - already proven in earlier discoveries - was chosen to complement a parallel copper-oxide chain. Then MnO2 (K=10,000) was selected to be the anion. This arrangement creates periodic compression of the oxygen ions positioned between the SnSbMn chains and the CuO chains.
|
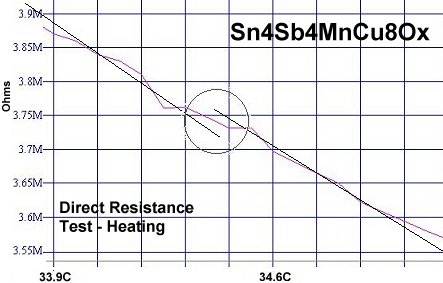
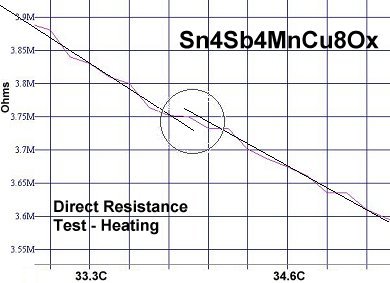
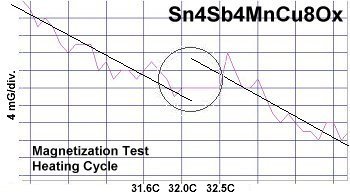
At page top are two direct resistance tests showing a sudden drop of about 20-30 kilohms near 34 Celsius (93F/307K) in this new compound. Though the volume fraction is low, the transitions appeared consistently at the same temperature1. Below are two magnetization tests showing Meissner transitions of about 8-10 milliGauss around 32-33 Celsius that confirm superconductivity. The signals were noisy due to the low volume fraction and high thermal noise figure inherent to these temperatures. Lines have been drawn through the noise to approximate the average of the data points. 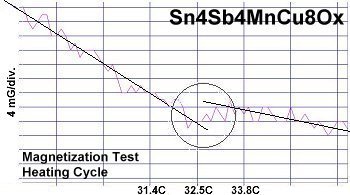
|
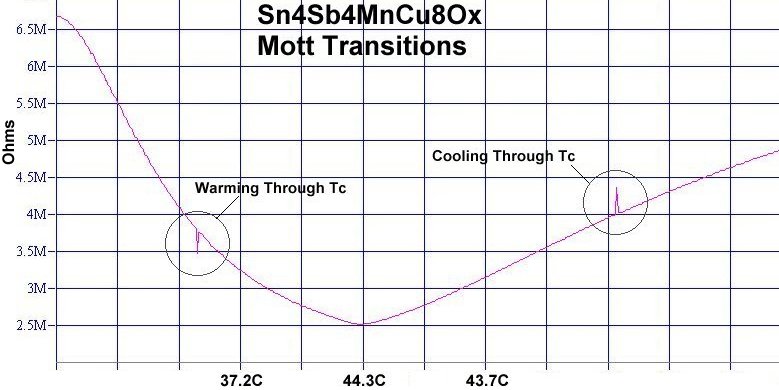
A third means of verifying superconductivity in this material is shown in the below plot. Since the non-superconductive host material is near a Mott transition, small transients are generated as the minority phase superconductor exits and later re-enters a superconductive state. As the pellet is warmed through Tc, the paired electrons are freed to flood the bulk, causing an over-doped state. This causes a momentary resistance drop. Then, as the material is cooled through Tc, electrons move to pair up, causing an under-doped condition. This causes a transient resistance increase. To read more about this phenomenon click here.
Stoichiometric ratios of the below chemicals were used to synthesize the test pellet:
SnO 99.9% (Alfa Aesar) 5.39 grainsThe chemical precursors were mixed and pelletized at 60,000 PSI and then sintered for 34 hours at 860C. Lastly the pellet was annealed for 10+ hours at 500C in flowing O2. Temperature was determined using an Omega type "T" thermocouple and precision OP77 DC amplifier. The magnetometer employed twin Honeywell SS94A1F Hall-effect sensors with a tandem sensitivity of 50 mv/Gauss.
RESEARCH NOTE: The copper-oxides are strongly hygroscopic. All tests should be performed immediately after annealing.
RE-PUBLICATION NOTICE: Elsevier Publishing, dba Elsevier Science, as well as Morris Communications, both print and broadcast divisions, are specifically prohibited from re-publishing any part of this news story.
E. Joe Eck
© 2015 Superconductors.ORG
All rights reserved.
1. Testing temperatures are believed accurate within ± 0.5 degrees C.
 BACK to "News" page at Superconductors.ORG
BACK to "News" page at Superconductors.ORG